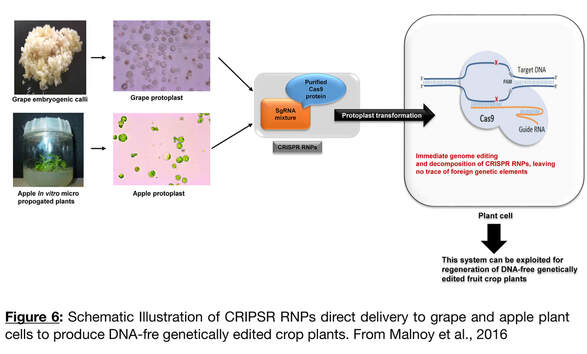
- Developing a Marker-Free Genome Editing methodology for clonally-propagated crop:
The use of embryogenic cells from which individual plants can be regenerated will address the first constraint. The use of Cell-Penetrating-Peptides to facilitate the penetration and the internalization of the RNPs could address the main issue of the delivering the cargo (Ramakrishna et al., 2014, Numata et al., 2018). We have developed a research project funded by the Wine Industry for a direct delivery of a SSN to edit grapevine material for key genes associated with performance traits. As a fisrt step, we aim to prove the feasibility of the direct delivery of a RNP into regenerable embryogenic cells.
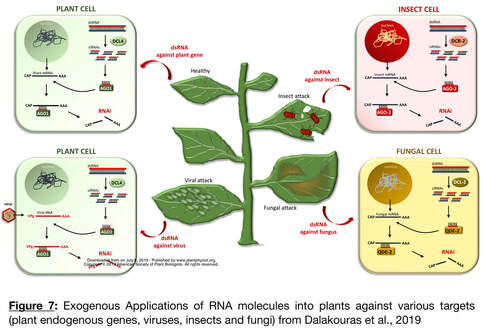
- Developing A Spray Induced Gene Silencing methodology for crop protection and improved fruit composition:
dsRNA molecules sprayed on the leaf surface were shown to be taken up by plant cells, transported to non-treated areas, and processed into sRNAs. These findings indicate that these applications can have systemic activity further enhancing their utility for pest and disease control. The premise is thus to deliver dsRNAs with a formulation made with clay particles using the same spray technology used for fungicides. Once applied, the dsRNA molecules complementary to target genes will disrupt the expression of the dsRNA-targeted genes in either the plant itself or the pathogens. The efficacy of SIGS was demonstrated for pest protection in multiple crops such as wheat, cucumber, pepper, etc.; and large-scale field trials are currently evaluated in Australia. The dsRNAs are reported to be stable and afford protection against the pathogen for up to 7-10 days, after which they degrade and leave no residue. The Fungus Erysiphe necator, responsible for Grape Powdery Mildew (GPM), is a significant threat to wine production that is mainly controlled using large quantities of fungicides. However, the emergence of fungicide resistance to chemicals suggests the need for rapid development of new strategies to control GPM. There is also a concern about the environmental impact of large-scale fungicide applications. Recent research demonstrates that agricultural pests, such as insects, nematodes, and fungi can be killed by exogenously supplied RNA molecules (Figure 7) opens opportunities to develop alternative field applications to control Powdery Mildew. We developed a SIGS to control GPM, we propose to accomplish three objectives.
In the first objective, we identified the stretches of RNA sequences with the maximum interference activity to silence fungal and native genes, EnCYP51, EnDCL1,2, and VitviMLOs 6,7, 11, respectively. In the second objective, we are currently evaluating the uptake and processing of RNA molecules by the grapevine and their systemic effects on the reduction of GPM. Finally, we will determine the efficacy of clay nanoparticles to stabilize and extend the lifespan of sprayed dsRNA molecules on grapevine leaves. Overall, we hope to provide the grape community with an RNAi-based spray technique for disease control. Our study will evaluate various aspects including the efficacy of silencing on the target genes, one adjuvant to use for long-lasting effects of dsRNA applications, the level of systemic effects, and the degree of PM-resistance afforded by the system in the laboratory studies. The results will be used for the development of field trials to evaluate the impact of SIGS system in reducing the cost of grapevine GPM management.
